
Drug Recall Law in India
Syllabus/GS2/Government Policies and Interventions for development in various sectors, Health
In News
- Recent failures of Indian manufactured drugs overseas (like the Gambia incident) have brought attention to the issue of absence of a drug recall law in the pharmaceutical industry in India.
What is a Drug Recall?
- Recall is an action taken to withdraw/remove the drugs from distribution or use including corrective action for which deficiencies are reported in quality, efficacy or safety.
- The defective products related to quality include Not of Standard Quality, Adulterated or Spurious drugs.
Status of Drug Recall Law in India
- India has been considering the creation of a mandatory recall law for substandard drugs since 1976, but still no law exists that mandates such medicine be removed from the market.
- In 1976, the Drugs Consultative Committee (DCC), discussed the issue of drug recalls with the various state regulators, however, none of them resulted in amendments to the Drugs & Cosmetics Act to create a mandatory recall mechanism.
- In 2012, the Central Drugs Standard Control Organisation (CDSCO) proposed a set of draft recall guidelines but the national regulator lacks the power to convert guidelines into a binding law.
- The issue of recalls resurfaced in 2016 at meetings of the DCC and at another committee, called the Drugs Technical Advisory Board (DTAB). Nothing changed on the ground. The same issue came up again in 2018 and 2019 at the meetings of the DCC, but India still lacks a recall law, 46 years on.
Reasons for not having the Law?
- Complex Drug Regulatory Issues: The Drug Regulation Section of the Union health ministry is simply not up to the task of tackling complex drug regulatory issues due to a combination of different factors including apathy, lack of expertise in the area, and a greater interest in enabling the growth of the pharmaceutical industry.
- No Centralised Regulatory Authority: India is a highly fragmented regulatory structure, with each state having its own drug regulator. Both the pharmaceutical industry and state drug regulators have resisted greater centralisation of regulatory powers.
- Fear of Public Scrutiny: Every month, dozens of drugs fail random-testing in government laboratories. Ideally, these drugs will be mandatorily recalled in a transparent manner, with the people being informed of the failures. But if this were to actually happen in India, the people at large would be flooded with alerts on an almost daily basis, which would then increase the pressure on drug regulators to institute extensive reforms.
Present Laws Regulating the Drugs and Pharmaceutical Sector in India
- Central Drugs Standard Control Organisation: It is the National Regulatory Authority (NRA) of India.
- The Drugs & Cosmetics Act,1940 and rules 1945 have entrusted various responsibilities to central & state regulators for regulation of drugs & cosmetics. It envisages uniform implementation of the provisions of the Act & Rules made there under for ensuring the safety, rights and well being of the patients by regulating the drugs and cosmetics.
- CDSCO along with state regulators, is jointly responsible for grant of licenses of certain specialized categories of critical Drugs such as blood and blood products, I. V. Fluids, Vaccine and Sera.

- The Drugs & Cosmetics Act, 1940: It regulates the import, manufacture, distribution and sale of drugs in India.
- The Pharmacy Act,1948: It is meant to regulate the profession of Pharmacy in India.
- The Drugs and Magic Remedies (Objectionable Advertisement) Act, 1954: It provides to control the advertisements regarding drugs; it prohibits the advertising of remedies alleged to possess magic qualities.
- The Narcotic Drugs and Psychotropic Substances Act, 1985: It is an act concerned with control and regulation of operations relating to Narcotic Drugs and Psychotropic Substances.
- National Pharmaceutical Pricing Authority: It is an organization of the Government of India which was established, inter alia, to fix/ revise the prices of controlled bulk drugs and formulations and to enforce prices and availability of the medicines in the country, under the Drugs (Prices Control) Order, 1995.
- The organization is also entrusted with the task of recovering amounts overcharged by manufacturers for the controlled drugs from the consumers. It also monitors the prices of decontrolled drugs in order to keep them at reasonable levels.
Way Ahead
- To create an effective recall mechanism, the responsibility of recalling drugs has to be centralised, with one authority that has the legal power to hold companies liable for failures to recall drugs from across the country, and further, to also search and seize batches of failed medicine.
- If India is a single market for drugs then it should have one regulator. If not, the incompetence of a regulator in just one state can lead to adverse effects for patients in other states, whose citizens have no influence or electoral power to demand accountability of that regulator.
Source: TH
Misleading Food Advertisements
Syllabus: GS2/ Government Policies & Interventions
In Context
- The Food Safety and Standards Authority of India (FSSAI) recently flagged 32 fresh cases of misleading ads and claims by Food Business Operators (FBOs).
More about the news
- In contravention of the Food Safety and Standards:
- The concerned advertisements were found to be in contravention of the Food Safety and Standards (Advertisements & Claims) Regulations, 2018.
- Who are these violators?
- FSSAI confirmed that they scrutinised products in varied categories such as health supplements, organic products, fast-moving consumer goods (FMCG) products and staples, each endorsing certain health and product claims.
- The alleged violators include manufacturers and/or marketers of nutraceutical products, refined oils, pulses, flours, millet products, and ghee.
Regulations
- Entity-specific regulations:
- FSSAI uses the Food Safety and Standards (Advertisements & Claims) Regulations, 2018 which specifically deal with food (and related products)
- Central Consumer Protection Authority (CCPA)’s regulations cover goods, products and services.
- Broadcasting regulations:
- The Programme and Advertising Codes prescribed under the Cable Television Network Rules, 1994 stipulate that advertisements must not draw inferences that it has “some special or miraculous or supernatural property or quality, which is difficult to be proved.”
- Consumer Protection Act, 2019:
- The Consumer Protection Act was enacted to deal with matters relating to violation of consumer’s rights, unfair trade practices, misleading advertisements, and all those circumstances which are prejudicial to the consumer’s rights.
- Prohibitions:
- Product claims suggesting suitability for prevention, alleviation, treatment or cure of a disease, disorder or particular psychological condition is prohibited unless specifically permitted under the regulations of the FSS Act, 2006.
- Punishments:
- Making deceptive claims or advertisements are punishable offences under Section-53 of the Food Safety and Standards Act, 2006.
Concerns & challenges
- Rising offences:
- As per the regulator, the cumulative count of such offences has shot up to 170 in the last six months.
- It urged FBOs to “desist from making any unscientific and/or exaggerated claims and advertisements to promote their product sales to avoid enforcement actions and in larger consumer interest.”
- As per the regulator, the cumulative count of such offences has shot up to 170 in the last six months.
- Technicalities:
- Most complaints of misleading were related to the nutrition of a product, its benefits and the ingredient mix not being based on adequate evidence. Issue here is that a lot of claim data is to be based on technical data.
- For example, if one says that there is Vitamin D in his/her product, we need evidence to substantiate that there indeed is Vitamin D in your product.
- Most complaints of misleading were related to the nutrition of a product, its benefits and the ingredient mix not being based on adequate evidence. Issue here is that a lot of claim data is to be based on technical data.
- Need of adequate clinical data:
- If the claim revolves around the composition of the product, saying that a product with all its elements achieves a certain outcome, companies need to provide clinical data about the outcomes pertaining to the control group, the administered group and the observed period of the claimed outcomes.
|
Related terminologies
|
Way Ahead
- What does the regulator expect?
- FSSAI seeks that the advertisements and claims be “truthful, unambiguous, meaningful, not misleading and help consumers to comprehend the information provided”.
- Claims must be scientifically substantiated by validated methods of characterising or quantifying the ingredient or substance that is the basis for the claim.
- It is extremely important for consumers to be able to distinguish between regular posts and promotional content.
- Any endorsement must reflect the genuine, reasonably current opinion of the individual, group or organisations making such representation and must be based on adequate information about, or experience with, the identified goods, product or service.
- An ideal advertisement can be suggestive and not passively imposing, it should educate the people and lastly, “it should be true!”
Source: TH
Thalassemia Bal Sewa Yojana
Syllabus: GS 2/Health
In News
- The Minister of State for Health launched the third phase of the Thalassemia Bal Sewa Yojana of the Health Ministry in New Delhi.
About Thalassemia Bal Sewa Yojana
- The Union Health Ministry has been implementing the Thalassemia Bal Sewa Yojana since 2017 under the National Health Mission (NHM) and recently completed its second phase in March 2023.
- The third phase of the program will continue to provide financial assistance for a package cost of 10 lakhs per Hematopoietic Stem Cell Transplant (HSCT), directly transferred from CIL to the institutions performing HSCT.
- It is being supported by Coal India Limited as part of their Corporate Social Responsibility initiative to commemorate World Thalassemia Day.
- It is a unique initiative aimed at providing a one-time cure opportunity to underprivileged Thalassemia patients who have a matched sibling donor but do not have the financial resources to cover the cost of the procedure.
Significance
- The program is an excellent example of a successful CSR initiative that is making a real difference in the lives of thalassemia patients and their families.
- The third phase will continue to bring hope and relief to thalassemia patients and their families and contribute towards building a healthier and more equitable society.
- The program will benefit underprivileged thalassemia patients and those with aplastic anemia, which is an irreversible condition that can be life-threatening without treatment
|
Thalassemia
Types of Thalassemia
|
Facts In News
Saint Samarth Ramdas
Syllabus/GS1/Famous Personalities
In News
- Recently a controversy irked after Jaggi Vasudev claimed that the Saint Samarth Ramdas was Chhatrapati Shivaji Maharaj’s guru.
Who was Samarth Ramdas?
- Samarth Ramdas (1608 – 1681), also known as Sant Ramdas or Ramdas Swami, was a Hindu saint, philosopher, poet, writer and spiritual master.
- He was a devotee of Lord Ram and Hanuman, he toured the entire Indian subcontinent for 12 years, during which he came across various spiritual teachers and diverse religious traditions.
- His paean to Lord Hanuman, Maruti Stotra, is still commonly recited by school children as well as wrestlers akhadas across Maharashtra.
Ramdasi sect
- He built a temple of Rama and Maruti (Hanuman) and founded a math or monastery which exists to the present day.
- The community of his followers soon expanded, taking on the character of a well marked sect, the Ramdasi sect, with its own sacred texts and forms of worship.
Philosophy
- Ramdas was an exponent of Bhakti Yoga or the path of devotion. According to him, total devotion to Rama brings about spiritual evolution.
- He expressed his admiration for warriors and highlighted their role in safeguarding the society. He was of the opinion that saints must not withdraw from society but instead actively engage towards social and moral transformation.
- He encouraged the participation of women in religious work and offered them positions of authority.
Legacy
- Ramdas has influenced Hindu nationalist thinkers across the years. The likes of Lokmanya Tilak, RSS founder KB Hedgewar, and VD Savarkar, all claimed to have been inspired by the 17th century saint.
Source: IE
Space Science and Technology AwaReness Training (START)
Syllabus: GS3/ Science & Technology, Space
In News
- The Indian Space Research Organisation (ISRO) has announced a new introductory-level online training programme called Space Science and Technology Awareness Training (START).
About the Programme
- Aim: To provide introductory-level training in Space Science and Technology to build human capacity for future Space Science and research.
- The programme will cover various domains of space science, including Astronomy & Astrophysics, Heliophysics & Sun-Earth interaction, Instrumentation, and Aeronomy.
- Participants: START is aimed at postgraduate and final-year undergraduate students of Physical Sciences and Technology.
- Implemented through: Jigyasa portal
About ISRO
- ISRO was previously the Indian National Committee for Space Research (INCOSPAR), set up by the Government of India in 1962, as envisioned by Dr. VikramA Sarabhai.
- ISRO was formed on August 15, 1969 and superseded INCOSPAR with an expanded role to harness space technology. DOS was set up and ISRO was brought under DOS in 1972.
- The prime objective of ISRO is the development and application of space technology for various national needs.
- HQ: Bengaluru
Source: TH
Project-SMART
Syllabus: GS 3/Infrastructure
In News
- The Ministry of Housing & Urban Affairs (MoHUA) has signed a memorandum of understanding (MoU) with the Japan International Cooperation Agency (JICA) and the Ministry of Railways for the Project-SMART
About Project-SMART
- It stands for Station Area Development along Mumbai-Ahmedabad High-Speed Rail(SMART).
- It is an initiative towards modernizing railway infrastructure in India, with the potential to transform the areas around the high-speed rail stations into bustling economic centers.
Features
- The MoU was inked for four high-speed stations — Sabarmati, Surat in Gujarat, and Virar and Thane in Maharashtra, out of the 12 stations in the route. Surat, Virar and
- Thane are green field projects while Sabarmati is a brown field development.
- The project — SMART envisages developing surrounding areas of MAHSR stations to enhance accessibility and convenience of commuters and other stakeholders as well as to promote economic activities in the vicinity of station areas.
Relevance
- It is an initiative towards modernizing railway infrastructure in India, with the potential to transform the areas around the high-speed rail stations into bustling economic centers.
- It will also enhance the institutional capacity of state governments, municipal corporations, and urban development authorities to plan, develop and manage the surrounding areas of MAHSR stations.
Source:TH
Respiratory Syncytial Virus (RSV)
Syllabus: GS3/ Science & Technology
In News
- In May 2023, the United States Food and Drug Administration (FDA) approved the Arexvy vaccine for respiratory syncytial virus (RSV).
Arexvy vaccine
- This is the first RSV vaccine to be approved anywhere in the world.
- Arexvy will lower respiratory tract disease in people older than 60 years.
- The vaccine is manufactured by the company, GSK.
Respiratory Syncytial Virus (RSV)
- RSV causes infections of the lungs and respiratory tract.
- Symptoms: RSV usually causes mild cold-like signs and symptoms such as Congested or runny nose, Dry cough, Low-grade fever, Sore throat, Sneezing and Headache.
- impact: In severe cases, RSV infection can lead to bronchiolitis (inflammation of the small airways in the lung) and pneumonia (infection of the lungs)
- Fatality: Lower respiratory tract disease caused by the RSV virus leads to approximately 60,000-1,20,000 hospitalisations and 6,000-10,000 deaths among adults who are 65 years of age and older.
- Vulnerable Groups: Older adults with comorbidities and infants are at high risk from RSV.
- Treatment: Most children and adults recover in one to two weeks, although some might require hospitalization.
- Protein F: Scientists have identified that when protein F, a fibronectin-binding protein, — which is yet to fully fuse with a human cell — is introduced in humans, it elicited neutralizing antibodies against the virus.
Source: TH
MiG-21 Crash
Syllabus: GS3/ Defence
In News
- A MiG-21 fighter jet of the Indian Air Force (IAF) crashed near Rajasthan’s Suratgarh. The exact cause of the accident is not known.
Status of the MIG-21 Jets in the IAF
- The MiG-21s are among the six fighter jets presently flown by the Indian Air Force (IAF).
- The Mikoyan-Gurevich MiG-21s are single engine, single-seater multi-role fighter/ground attack aircraft designed by the Mikoyan-Gurevich Design Bureau in the Soviet Union/Russia.
- They were first inducted by India in 1963. India has procured over 700 MiG-21 aircraft of different variants since then, such as the Type-77, Type-96 and the BIS. The latest of them are the MiG-21 Bison.
- Role: The aircraft in the past had proved their mettle in several wars (1965, 1971 and 1999 Kargil conflict) fought by India against Pakistan.
MiG accidents
- Incidents: As per available government data, close to 500 MiG-21s have crashed in the last 60 years, killing over 170 pilots.
- Causes: The reasons for the crashes have been several — ranging from technical defects, human error, bird hits or spatial disorientation of pilots in certain circumstances.
Phasing out of MiG
- In September 2022, the No. 51 squadron was number plated.
- The three MiG-21 Bison squadrons are planned to be phased out by December 2025.
- They will be replaced by the indigenous Light Combat Aircraft Tejas squadrons.

Source: IE
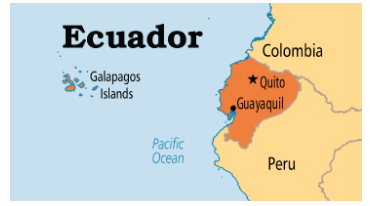
Galapagos Islands
Syllabus: GS1/ Places in News
In News
- Credit Suisse, the Swiss bank, has announced buying Ecuadorian bonds worth $1.6 billion in a debt-for-nature swap that cost the Swiss bank just $644 million.
- In return, Ecuador’s government will have to spend about $18 million annually for two decades on conservation in the Galapagos Islands.
About
- The Galapagos Islands are an archipelago of volcanic islands belonging to south American country ‘Ecuador’.
- Location: The archipelago is located in the Eastern Pacific around the Equator. Straddling the equator, islands in the chain are located in both the northern and southern hemispheres.
- Geology: The archipelago is located on the Nazca Plate (a tectonic plate), which is moving east/southeast, diving under the South American Plate. Volcanism has been continuous on the Galápagos Islands for at least 20 million years.
- Ecology: The Galapagos are famous for their large number of endemic species, which were studied by Charles Darwin in the 1830s and inspired his theory of evolution by means of natural selection. The Galapagos Islands are named after their giant tortoises.
Source: IE